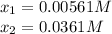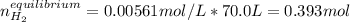Chemistry, 22.05.2020 02:10 Flameking1223
For the reaction
H2(g) + CO2(g) ⇆ H2O(g) + CO(g)
at 700°C, Kc = 0.534. Calculate the number of moles of H2 that are present at equilibrium if a mixture of 0.680 mole of CO and 0.680 mole of H2O is heated to 700°C in a 70.0−L container.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 00:00
Draw the skeletal structures of two different molecules that are each made of 5 carbon atoms and 12 hydrogen atoms.
Answers: 1

Chemistry, 22.06.2019 00:30
What does x represent in the formula for the compound xcl4?
Answers: 2

Chemistry, 22.06.2019 04:30
When the water vapor cools it condenses select a number that represents his process on the
Answers: 3

Chemistry, 22.06.2019 07:50
In which situation can a mixture always be called a solution
Answers: 3
You know the right answer?
For the reaction
H2(g) + CO2(g) ⇆ H2O(g) + CO(g)
at 700°C, Kc = 0.534. Calcu...
H2(g) + CO2(g) ⇆ H2O(g) + CO(g)
at 700°C, Kc = 0.534. Calcu...
Questions









French, 19.05.2021 01:10

Mathematics, 19.05.2021 01:10


Mathematics, 19.05.2021 01:10




Mathematics, 19.05.2021 01:10

Mathematics, 19.05.2021 01:10



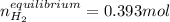
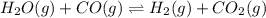
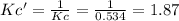
![Kc'=\frac{[H_2][CO_2]}{[H_2O][CO]}](/tpl/images/0660/7360/14ed3.png)
 due to the reaction extent:
due to the reaction extent:![Kc'=\frac{(x)(x)}{([H_2O]_0-x)([CO]_0-x)}=1.87](/tpl/images/0660/7360/2d81b.png)
![[H_2O]_0=[CO]_0=\frac{0.680mol}{70.0L}=0.0097M](/tpl/images/0660/7360/92f9f.png)