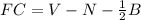Chemistry, 20.04.2020 21:15 victordhernandez01
Due to the small and highly electronegative nature of fluorine, the oxyacids of the this element are much less common and less stable than those of the other halogens. Bonding theory, however, does allow one to propose structures for these acids and use formal charges for the evaluation of these structures. For a molecule of fluorous acid, the atoms are arranged as HOFO. (Note: In this oxyacid, the placement of fluorine is an exception to the rule of putting the more electronegative atom in a terminal position.) What is the formal charge on each of the atoms?

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 18:40
What kind of ion is contained in salts that produce an acidic solution? a positive ion that attracts a proton from water a positive ion that releases a proton to water a negative ion that attracts a proton from water a negative ion that releases a proton to water
Answers: 1

Chemistry, 22.06.2019 06:40
Ted and emily played a mixed doubles tennis match against jack and brenda. in the second match. ted and brenda played against jack and emily. which type of chemical reaction does the situation demonstrate?
Answers: 3

Chemistry, 22.06.2019 10:30
Acompound has a molar mass of 92.02 grams/mole, and its percent composition is 30.4% nitrogen (n) and 69.6% oxygen (o). what is its molecular formula? a. n2o4 b. no2 c. n2o d. n4o2
Answers: 1

Chemistry, 22.06.2019 19:30
Acetylene gas c2h2 undergoes combustion to produce carbon dioxide and water vapor how many grams of water are produced by the same amount of c2h2?
Answers: 2
You know the right answer?
Due to the small and highly electronegative nature of fluorine, the oxyacids of the this element are...
Questions



English, 04.02.2021 01:00

Biology, 04.02.2021 01:00

Social Studies, 04.02.2021 01:00

History, 04.02.2021 01:00

Biology, 04.02.2021 01:00

Mathematics, 04.02.2021 01:00

Business, 04.02.2021 01:00


Social Studies, 04.02.2021 01:00

Mathematics, 04.02.2021 01:00


Geography, 04.02.2021 01:00


Social Studies, 04.02.2021 01:00



English, 04.02.2021 01:00