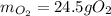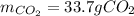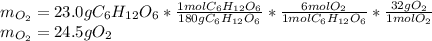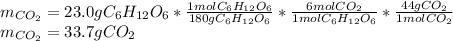Glucose, C6H12O6, is used as an energy source by the human body. The overall reaction in the body is described by the equation:
C6H12O6(aq) + 6O2(g) ⟶ 6CO2(g) + 6H2O(l)
1) Calculate the number of grams of oxygen required to convert 23.0 g of glucose to CO2 and H2O.
2) Calculate the number of grams of CO2 produced.

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 09:20
Explain that newton first law,second law and third law of motion?
Answers: 2


Chemistry, 22.06.2019 16:00
Which factor is likely to impact the possible number of compounds ?
Answers: 1
You know the right answer?
Glucose, C6H12O6, is used as an energy source by the human body. The overall reaction in the body is...
Questions

History, 03.03.2020 03:30




Mathematics, 03.03.2020 03:30



Geography, 03.03.2020 03:30



History, 03.03.2020 03:30

Computers and Technology, 03.03.2020 03:30

Biology, 03.03.2020 03:30






History, 03.03.2020 03:30

Biology, 03.03.2020 03:30