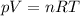What is the molar mass of a gas with a density of 3.50 g/L at a
temperature of 345 K and a pre...

Chemistry, 05.04.2020 22:22 raymondanthony3314
What is the molar mass of a gas with a density of 3.50 g/L at a
temperature of 345 K and a pressure of 0.950 atm? (Write your
answer whole number e. g. 345)
g/mol

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 12:30
Avariable that is not being directly tested during an experiment should be
Answers: 1

Chemistry, 22.06.2019 18:00
Alidded glass container is filled with a colored gas. after a period of time, it is observed that the gas is uniformly spread throughout the box and that the movement has slowed considerably. next, a warm iron plate is carefully placed under the box. why is there resumed movement of the gas in the container?
Answers: 2


Chemistry, 22.06.2019 21:30
Isopropyl alcohol, (ch3)2choh, is a common solvent. determine the percent by mass of hydrogen in isopropyl alcohol. a) 6.71% h b) 13.4% h c) 25.0% h d) 53.3% h
Answers: 1
You know the right answer?
Questions