Note the average atomic mass of copper on the nano-balance. Add atoms to the
larger balance un...

Note the average atomic mass of copper on the nano-balance. Add atoms to the
larger balance until it registers the same number in g) as the reading on the nano-balance
(in u). Use the Exponent slider to help get the correct amount. Stop adding atoms when the
readings on both balances matchi exactly to the nearest 0.001 y).
How many atoms did you need to add?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 18:20
Categorize them by metal, nonmetal, in periodic tableductilenon-ductilemalleableoften gain electrons easilygood conductorpoor conductorcan be liquidselements
Answers: 2

Chemistry, 22.06.2019 23:00
Consider the reaction: 2al(s) + fe2o3(s) → al2o3(s) + 2fe(s) the δhf for fe2o3(s) = -824.3 kj/mole. the δhf for al2o3(s) = -1675.7 kj/mole. finish the equation. δhrxn = [(1)( kj/mole) + (2)( kj/mole)] - [(1)( kj/mole) + (2) ( kj/mole)]
Answers: 1

Chemistry, 22.06.2019 23:30
What are the similarities between compounds and mixtures?
Answers: 3

You know the right answer?
Questions
















Computers and Technology, 29.11.2020 15:10




 1
1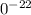 g
.
g
.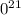 atoms.
atoms.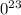 atoms or molecules of that substance - this is known as Avogadro's number.
atoms or molecules of that substance - this is known as Avogadro's number.

