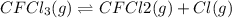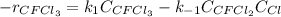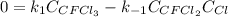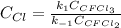Chemistry, 20.03.2020 10:54 diwashkandel6pe02af
Consider the following elementary reaction: CFC13(g)-CFC12(9)+Clg) Suppose we let k1 stand for the rate constant of this reaction, and k1 stand for the rate constant of the reverse reaction Write an expression that gives the equilibrium concentration of Cl in terms of k, k_1, and the equilibrium concentrations of CFCI3 and CFCI2 1. K-1 [ci]

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 03:30
The boiling point of liquids is very high what does it indicate
Answers: 1

Chemistry, 22.06.2019 07:20
Which of these conditions most likely produces an unstable isotope?
Answers: 1

Chemistry, 22.06.2019 13:30
Which is true of a liquid? it has a definite volume but not a definite mass.it has a definite mass but not a definite volume.it has a definite volume but not a definite shape.it has a definite shape but not a definite volume.
Answers: 2

Chemistry, 22.06.2019 13:50
What happens when an atom of sulfur combines with two atoms of chlorine to produce sci2? a. each chlorine atom shares a pair of electrons with the sulfur atom. b. an electron is transferred from each chlorine atom to the sulfur atom. c. an electron is transferred from the sulfur atom to each chlorine atom. d. each chlorine atom shares all its valence electrons with the sulfur atom.
Answers: 2
You know the right answer?
Consider the following elementary reaction: CFC13(g)-CFC12(9)+Clg) Suppose we let k1 stand for the r...
Questions

English, 25.10.2019 06:43



Mathematics, 25.10.2019 06:43


Mathematics, 25.10.2019 06:43


Health, 25.10.2019 06:43

Physics, 25.10.2019 06:43







Mathematics, 25.10.2019 06:43


Mathematics, 25.10.2019 06:43


Mathematics, 25.10.2019 06:43

![[Cl]_{eq}=\frac{k_1[CFCl_3]_{eq}}{k_{-1}[CFCl_2]_{eq}}](/tpl/images/0555/9861/640b9.png)