
Chemistry, 11.03.2020 22:56 amberpetty4288
The value of Ka for acetic acid , CH3COOH , is 1.80×10-5 . Write the equation for the reaction that goes with this equilibrium constant. (Use H3O+ instead of H+.)

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 01:20
1. suppose a reaction mixture, when diluted with water, afforded 300 ml of an aqueous solution of 30 g of the reaction product malononitrile [ch2(cn)2], which is to be isolated by extraction with ether. the solubility of malononitrile in ether at room temperature is 20.0 g/100 ml, and in water is 13.3 g/100 ml. what weight of malononitrile would be recovered by extraction with (a) three 100-ml portions of ether and (b) one 300-ml portion of ether? suggestion: for each extraction, let x equal the weight extracted into the ether layer. in part (a), the concentration in the ether layer is x/100 and in the water layer is (30 x)/300; the ratio of these quantities is equal to k 20/13.3.
Answers: 2

Chemistry, 22.06.2019 02:30
When you perform this reaction, what could remain at the end of the reaction? check all that apply. excess reactant aqueous copper chloride excess reactant aluminum oxygen product solid copper carbon dioxide product aqueous aluminum chloride water
Answers: 2


Chemistry, 22.06.2019 19:30
Anurse used a 0.02-mg/l solution of disinfection to clean a patients wound. what is the concentration of the solution expressed as a percentage?
Answers: 1
You know the right answer?
The value of Ka for acetic acid , CH3COOH , is 1.80×10-5 . Write the equation for the reaction that...
Questions

Mathematics, 05.03.2021 22:30

Mathematics, 05.03.2021 22:30

Mathematics, 05.03.2021 22:30


Mathematics, 05.03.2021 22:30





Social Studies, 05.03.2021 22:30






Mathematics, 05.03.2021 22:30


Mathematics, 05.03.2021 22:30

Biology, 05.03.2021 22:30

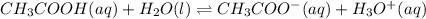
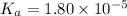
![K_c=\frac{[CH_3COO^-][H_3O^+]}{[CH-3COOH][H_2O]}](/tpl/images/0543/5879/7c275.png)
![K_a=K_c\times [H_2O]=\frac{[CH_3COO^-][H_3O^+]}{[CH-3COOH]}](/tpl/images/0543/5879/125a5.png)
![[H_2O]=1](/tpl/images/0543/5879/b8579.png)
![K_a=\frac{[CH_3COO^-][H_3O^+]}{[CH-3COOH]}](/tpl/images/0543/5879/e4df0.png)


