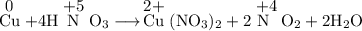Chemistry, 11.03.2020 22:23 gudtavosanchez19
Consider the reaction that occurs when copper is added to nitric acid. Cu(s)+4HNO3(aq) > Cu(NO3)2(aq)+2NO2(g)+2H2O(I). What is the reducing agent in this reaction? Cu NO3– Cu(NO3)2 NO2

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 09:20
How have the greenhouse gasses increased from the year 2000 to 2018
Answers: 2



Chemistry, 22.06.2019 20:00
Nitrogen dioxide decomposes according to the reaction 2 no2(g) ⇌ 2 no(g) + o2(g) where kp = 4.48 × 10−13 at a certain temperature. if 0.70 atm of no2 is added to a container and allowed to come to equilibrium, what are the equilibrium partial pressures of no(g) and o2(g)
Answers: 2
You know the right answer?
Consider the reaction that occurs when copper is added to nitric acid. Cu(s)+4HNO3(aq) > Cu(NO3)2...
Questions


Mathematics, 30.10.2020 03:40




Biology, 30.10.2020 03:40

Mathematics, 30.10.2020 03:40

Mathematics, 30.10.2020 03:40


Engineering, 30.10.2020 03:40

Mathematics, 30.10.2020 03:40


Mathematics, 30.10.2020 03:40



English, 30.10.2020 03:40

Mathematics, 30.10.2020 03:40



Mathematics, 30.10.2020 03:40