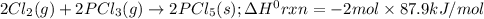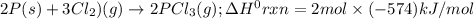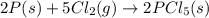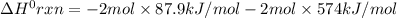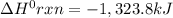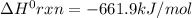Chemistry, 11.03.2020 06:31 cyanezc1313
Consider the following thermochemical equations. PCl5 (s)→PCl3 (g)+Cl2 (g)2P (s)+3Cl2 (g)→2PCl3 (g)ΔH∘rxn=87.9kJmol ΔH∘rxn=−574kJmol Using this data, determine the heat of formation for PCl5.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 04:30
Acamcorder has a power rating of 17 watts. if the output voltage from its battery is 7 volts, what current does it use?units:
Answers: 1

Chemistry, 22.06.2019 11:00
Which are examples of how technology has advanced scientific understanding.1using hot water to sterilize medical equipment.2transplanting a human organ into another individual.3inserting genes from one sheep into another cell to make a cloneunderstanding the different structures that make up a cell.4examining microorganisms from the deepest parts of the ocean
Answers: 2

Chemistry, 22.06.2019 12:10
Consider the reaction: n2(g) + o2(g) ⇄ 2no(g) kc = 0.10 at 2000oc starting with initial concentrations of 0.040 mol/l of n2 and 0.040 mol/l of o2, calculate the equilibrium concentration of no in mol/l how would this be done?
Answers: 3

You know the right answer?
Consider the following thermochemical equations. PCl5 (s)→PCl3 (g)+Cl2 (g)2P (s)+3Cl2 (g)→2PCl3 (g)Δ...
Questions




Mathematics, 12.01.2020 02:31

Mathematics, 12.01.2020 02:31

Physics, 12.01.2020 02:31



Mathematics, 12.01.2020 02:31






Mathematics, 12.01.2020 02:31

Mathematics, 12.01.2020 02:31

Mathematics, 12.01.2020 02:31

History, 12.01.2020 02:31

Mathematics, 12.01.2020 02:31

English, 12.01.2020 02:31