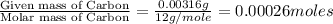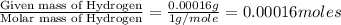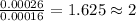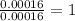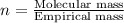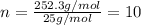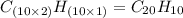Chemistry, 10.03.2020 09:06 auviannadority13
One of the components of natural crude oil and coal deposits is benzo[a]pyrene, a compound with a molecular mass of about 252.3 amu, containing only carbon and hydrogen. A 3.320 mg sample of benzo[a]pyrene burns to give 11.58 mg of CO2. Determine its empirical and molecular formulas. (Omit states-of-matter from your answer.)

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 04:20
Which of the following is true for the actual yield of a reaction? it is always calculated as a ratio. it is the yield from the excess reactant. it is the yield from the limiting reactant. it is always less than the theoretical yield.
Answers: 1

Chemistry, 22.06.2019 09:10
How have the greenhouse gasses increased from the year 2000 to 2018
Answers: 2

Chemistry, 22.06.2019 11:30
Determine the reaction and balance the following equations urgent due in the morning
Answers: 2
You know the right answer?
One of the components of natural crude oil and coal deposits is benzo[a]pyrene, a compound with a mo...
Questions


Mathematics, 04.02.2020 20:48

Biology, 04.02.2020 20:48

Biology, 04.02.2020 20:48



English, 04.02.2020 20:48

Mathematics, 04.02.2020 20:48




English, 04.02.2020 20:48



French, 04.02.2020 20:48



World Languages, 04.02.2020 20:48


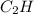 and
and 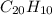
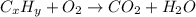
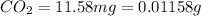 (Conversion factor: 1 g = 1000 mg)
(Conversion factor: 1 g = 1000 mg)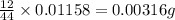 of carbon will be contained.
of carbon will be contained.