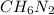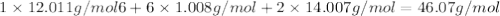Chemistry, 02.03.2020 07:15 snowprincess99447
Determine the molecular formula for the compound with a molar mass of 46.08 g/mol and the following percent compostion:
C:H:N:26.06%13.13%60.81%

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 12:30
Estructura 7.2 completar repaso 1 - ¿lógico o ilógico? 1 - ¿lógico o ilógico? listen and indicate whether each question and response is lógico or ilógico.
Answers: 3

Chemistry, 22.06.2019 11:00
The diagram below shows the different phase transitions that occur in matter. which arrow represents the transition in which dew is formed?
Answers: 1

Chemistry, 23.06.2019 01:30
Magnesium is the limiting reactant in this experiment. calculate the theoretical yield of mgo for each trial. trial 1: trial 2: data mass of empty crucible with lid trial 1: 26.688 trial 2: 26.681 mass of mg metal, crucible, and lid trial 1: 26.994 trial: 2 26.985 mass of mgo, crucible, and lid trial 1: 27.188 trial 2: 27.180
Answers: 1

Chemistry, 23.06.2019 04:40
Equal numbers of moles of he(g), ar(g), and ne(g) are placed in a glass vessel at room temperature. if the vessel has a pinhole-sized leak, which of the following will be true regarding the relative values of the partial pressures of the gases remaining in the vessel after some of the gas mixture has effused?
Answers: 1
You know the right answer?
Determine the molecular formula for the compound with a molar mass of 46.08 g/mol and the following...
Questions

English, 25.01.2021 01:00


Mathematics, 25.01.2021 01:00



Physics, 25.01.2021 01:00


Social Studies, 25.01.2021 01:00





History, 25.01.2021 01:00





Mathematics, 25.01.2021 01:00

Mathematics, 25.01.2021 01:00