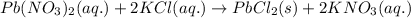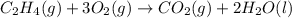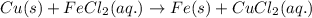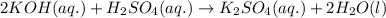Classify each of these reactions. Pb ( NO 3 ) 2 ( aq ) + 2 KCl ( aq ) ⟶ PbCl 2 ( s ) + 2 KNO 3 ( aq ) C 2 H 4 ( g ) + 3 O 2 ( g ) ⟶ 2 CO 2 ( g ) + 2 H 2 O ( l ) Cu ( s ) + FeCl 2 ( aq ) ⟶ Fe ( s ) + CuCl 2 ( aq ) 2 KOH ( aq ) + H 2 SO 4 ( aq ) ⟶ K 2 SO 4 ( aq ) + 2 H 2 O ( l )

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 18:20
Which statement accurately describes the relationship between air pressure, air density, or altitude? as altitude increases, pressure increases.as altitude increases, air density increases.air pressure and density are lowest at sea level.denser air exerts more pressure than less dense air.
Answers: 2

Chemistry, 21.06.2019 22:30
Consider the following system at equilibrium. caco3(s) ca2+(aq) + co32–(aq) the addition of which compound will cause a shift in equilibrium because of a common ion effect? ccl4 co2 cuso4 na2co3
Answers: 3

Chemistry, 22.06.2019 12:40
When 13.3 g koh is dissolved in 102.7 g of water in a coffee-cup calorimeter, the temperature rises from 21.4 °c to 31.53 °c. what is the enthalpy change per gram of koh (j/g) dissolved in the water? * take the density of water as 1.00 g/ml. * assume that the solution has a specific heat capacity of 4.18 j/g*k. enter to 1 decimal place. do not forget the appropriate sign /(+). canvas may auto-delete the (+) sign
Answers: 2

Chemistry, 22.06.2019 17:30
Consider the story you just read. all but one of the choices below indicate that something is living.
Answers: 1
You know the right answer?
Classify each of these reactions. Pb ( NO 3 ) 2 ( aq ) + 2 KCl ( aq ) ⟶ PbCl 2 ( s ) + 2 KNO 3 ( aq...
Questions

Biology, 19.11.2019 23:31










Computers and Technology, 19.11.2019 23:31






Mathematics, 19.11.2019 23:31