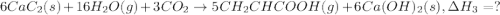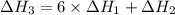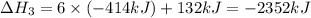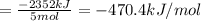Chemistry, 19.11.2019 23:31 sipstick9411
There are two steps in the usual industrial preparation of acrylic acid, the immediate precursor of several useful plastics. in the first step, calcium carbide and water react to form acetylene and calcium hydroxide: (s) (g) (g) (s) in the second step, acetylene, carbon dioxide and water react to form acrylic acid: (g) (g) (g) (g) calculate the net change in enthalpy for the formation of one mole of acrylic acid from calcium carbide, water and carbon dioxide from these reactions. round your answer to the nearest .

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 20:30
18. use the activity series to predict whether the following synthesis reaction will occur. write the chemical equations for the reaction if it's predicted to occur. (s) + o2(g) -> *note: it is possible.*
Answers: 1

Chemistry, 22.06.2019 06:00
Match the name of the following compound: mgso4 · h2omagnesium sulfate monohydratemagnesium (ii) sulfate monohydratemagnesium (ii) sulfate hydratemagnesium sulfate hydrate
Answers: 1

Chemistry, 22.06.2019 08:30
Which of the following would be an accurate picture of the earth during the summer time of the northern hemisphere?
Answers: 1

Chemistry, 22.06.2019 09:10
How have the greenhouse gasses increased from the year 2000 to 2018
Answers: 2
You know the right answer?
There are two steps in the usual industrial preparation of acrylic acid, the immediate precursor of...
Questions



Chemistry, 02.11.2020 02:30

Advanced Placement (AP), 02.11.2020 02:30

English, 02.11.2020 02:30



Spanish, 02.11.2020 02:30


Geography, 02.11.2020 02:30

History, 02.11.2020 02:30

Physics, 02.11.2020 02:30





Physics, 02.11.2020 02:30


Social Studies, 02.11.2020 02:30

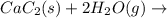
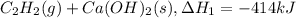 ..[1]
..[1]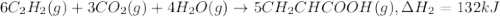 ..[2]
..[2]