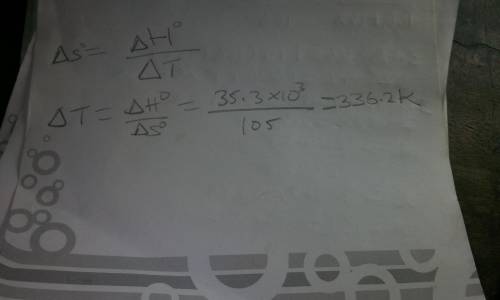Chemistry, 12.02.2020 00:32 nommies005
What is the boiling point of methanol, if ∆H°for vaporization is 35.3 kJ/mol, and ∆S° for vaporization is 105 J/mol K?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 13:00
How many moles of sulfur dioxide are produced when 4.38 moles of oxygen completely react with iron (iv) sulfide
Answers: 2


Chemistry, 23.06.2019 06:20
An object of mass 10.0 kg and volume 1000 ml and density 10 g/ml sinks in water who’s density is 1.0 g/ml. what is the mass of the water which has been displaced in kilograms
Answers: 1

Chemistry, 23.06.2019 07:00
Under what conditions will a gas be most likely to exhibit the ideal gas properties predicted by the ideal gas law? 1)high pressures and high temperature, because particles are forced closer together with higher kinetic energy, so intermolecular forces of attraction are weaker 2)high pressure and low temperature, because particles are forced closer together and moving slower, so the volume of the particles is less significant 3) low pressure and high temperature, because particles are spread farther apart and moving faster, so the intermolecular forces of attraction are weaker 4)low pressure and low temperature, because particles are spread farther apart with lower kinetic energy, so the volume of the particles is less significant
Answers: 2
You know the right answer?
What is the boiling point of methanol, if ∆H°for vaporization is 35.3 kJ/mol, and ∆S° for vaporizati...
Questions

Mathematics, 19.03.2020 21:38



Mathematics, 19.03.2020 21:38






Mathematics, 19.03.2020 21:38

Mathematics, 19.03.2020 21:38


History, 19.03.2020 21:38

Mathematics, 19.03.2020 21:38


History, 19.03.2020 21:38

Mathematics, 19.03.2020 21:38

English, 19.03.2020 21:38