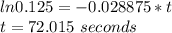The decomposition of formic acid follows first-order kinetics. HCO2H(g) → CO2(g) + H2(g) The half-life for the reaction at 550°C is 24 seconds. How many seconds does it take for the formic acid concentration to decrease by 87.5%? (1) 4.6 seconds (2) 36 seconds (3) 48 seconds (4) 72 seconds (5) 96 seconds

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 02:50
What is the overall order of reaction for rate = k[no2]2 ? second order 3/2 order third order zero order none of the listed answers are correct
Answers: 3

Chemistry, 22.06.2019 05:30
Which of the following signs of a chemical reaction are observed in the reaction of potassium with water? precipitate formed temperature change smell produced gas produced color change
Answers: 2

Chemistry, 22.06.2019 10:00
Select all of the methods through which a drug can enter your body. injection swallowing inhalation absorption
Answers: 2

You know the right answer?
The decomposition of formic acid follows first-order kinetics. HCO2H(g) → CO2(g) + H2(g) The half-li...
Questions





SAT, 25.11.2021 14:10

SAT, 25.11.2021 14:10



Mathematics, 25.11.2021 14:10

Mathematics, 25.11.2021 14:10





English, 25.11.2021 14:10



Chemistry, 25.11.2021 14:10

Social Studies, 25.11.2021 14:10

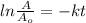
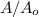 will become 0.125
will become 0.125