Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 20:00
For the reaction c6h14(g) & longrightarrow; c6h6(g) + 4h2(g), δp(h2)/δt was found to be 2.5 x 10-2 atm/s, where δp(h2) is the change in pressure of hydrogen. determine δp(c6h14)/δt for this reaction at the same time.
Answers: 2

Chemistry, 23.06.2019 10:30
Describe the hybridization of each carbon and nitrogen atom in each of the following structures
Answers: 1


Chemistry, 23.06.2019 13:30
If a fast moving car making a loud noise approaches and moves past the person what will happen as the distance between the two increases?
Answers: 1
You know the right answer?
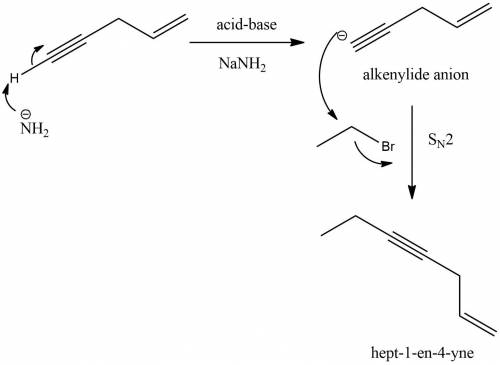
Predict the major product(s) for the reaction of pent-1-en-4-yne with sodium amide followed by react...
Questions

Mathematics, 11.07.2019 11:10





History, 11.07.2019 11:20


History, 11.07.2019 11:20

Mathematics, 11.07.2019 11:20




History, 11.07.2019 11:20

History, 11.07.2019 11:20



History, 11.07.2019 11:20


Mathematics, 11.07.2019 11:20

Biology, 11.07.2019 11:20

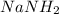 ) gives acid-base reaction with terminal alkyne preferably than terminl alkene. Because terminal alkyne are more acidic due to sp-hybridization of carbon atom.
) gives acid-base reaction with terminal alkyne preferably than terminl alkene. Because terminal alkyne are more acidic due to sp-hybridization of carbon atom.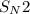 reaction with bromoethane to produce hept-1-en-4-yne.
reaction with bromoethane to produce hept-1-en-4-yne.