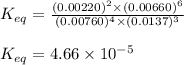9. the first step in industrial nitric acid production is the catalyzed oxidation of ammonia. without a catalyst, a different reaction predominates: 4nh3(g) + 3o2(g) ⇔ 2n2(g) + 6h2o(g) when 0.0120 mol gaseous nh3 and 0.0170 mol gaseous o2 are placed in a 1.00 l container at a certain temperature, the n2 concentration at equilibrium is 2.20×10-3 m. calculate keq for the reaction at this temperature.

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 14:00
How many absorptions would you expect to observe in the 13c nmr spectra of the following molecules? a) 3-chloropentane b) cis-4-methyl-2-pentene
Answers: 2

Chemistry, 22.06.2019 14:00
How is the atomic number of a nucleus changed by alpha decay
Answers: 2

Chemistry, 22.06.2019 17:40
Areaction in which products can react to re-form reactants is
Answers: 1
You know the right answer?
9. the first step in industrial nitric acid production is the catalyzed oxidation of ammonia. withou...
Questions

Geography, 03.12.2019 01:31

Mathematics, 03.12.2019 01:31



Biology, 03.12.2019 01:31

Mathematics, 03.12.2019 01:31

Mathematics, 03.12.2019 01:31

Mathematics, 03.12.2019 01:31

Arts, 03.12.2019 01:31


Physics, 03.12.2019 01:31


Social Studies, 03.12.2019 01:31


Mathematics, 03.12.2019 01:31




Mathematics, 03.12.2019 01:31

Mathematics, 03.12.2019 01:31

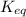 is
is 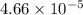
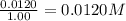
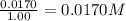
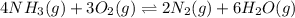
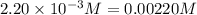
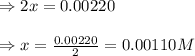
![0.0120-4x=[0.0120-(4\times 0.00110)]=0.00760M](/tpl/images/0421/4577/5e570.png)
![0.0170-3x=[0.0170-(3\times 0.00110)]=0.0137M](/tpl/images/0421/4577/b5ac6.png)
![6x=[6\times 0.00110]=0.00660M](/tpl/images/0421/4577/d0282.png)
![K_{eq}=\frac{[N_2]^2\times [H_2O]^6}{[NH_3]^4\times [O_2]^3}](/tpl/images/0421/4577/7f887.png)