
Chemistry, 26.11.2019 23:31 sarahlearn3
Lithium and fluorine undergo ionic bonding. using the noble gas electron configurations for each (below), explain the process of bonding step by step, using proper grammar and mechanics. noble gas electron configurations: lithium: [he] 2s1 fluorine: [he] 2s22p5

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 12:30
Avariable that is not being directly tested during an experiment should be changed varied experimented controlled
Answers: 1


Chemistry, 22.06.2019 14:30
1) describe the physical layout of the ocean floor ? 2) explain how the dumbo octopus swims differently than other octopus species and why this would be an advantage in the aphonic zone . 3) why are the types of organisms that live at each underwater hot vent so dramatically different ?
Answers: 3
You know the right answer?
Lithium and fluorine undergo ionic bonding. using the noble gas electron configurations for each (be...
Questions




History, 03.05.2021 05:50

History, 03.05.2021 05:50


Mathematics, 03.05.2021 05:50



Mathematics, 03.05.2021 05:50




Mathematics, 03.05.2021 05:50


English, 03.05.2021 05:50



Mathematics, 03.05.2021 05:50

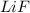 .
.![[He]2s^1](/tpl/images/0392/2650/6e198.png) .
.
![[He]2s^22p^5](/tpl/images/0392/2650/da8fe.png) .
.



