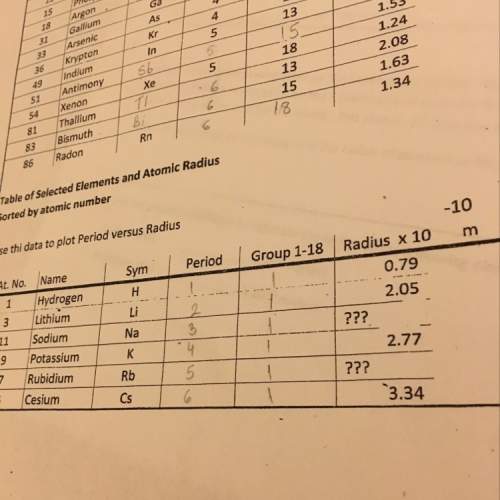
Chemistry, 04.12.2019 10:31 erikap0889
The net energy released or absorbed during a reversible chemical reaction is equal to
(1) the activation energy of the endothermic reaction
(2) the activation energy of the exothermic reaction
(3) the difference between the potential energy of the products and the potential energy of the reactants
(4) the sum of the potential energy of the products and the potential energy of the reactants

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 22:20
Which of the following statements is false regarding aromaticity? a. the compound must be cyclic b. the compound must be fully conjugated c. the compound must be planar d.the number of electrons in the pi system must satisfy the hückel 4n+2 rule e. the compound must have a neutral charge
Answers: 2

Chemistry, 22.06.2019 05:00
Given sno2 + 2h2 - sn + 2h20 tin oxide reacts with hydrogen to produce tin and water. how many moles of sno2 are needed to produce 500.0 grams of sn?
Answers: 3


Chemistry, 22.06.2019 18:00
Mercury turns to vapor at 629.88 k how much heat is lost 175 g of mercury vapor at 650 current condenses to a liquid at 297 ca mercury turns to weber at 629.88 kelvin how much he is lost 175 g of mercury vapor and 650 coming condensers to liquidate 297 kevin
Answers: 2
You know the right answer?
The net energy released or absorbed during a reversible chemical reaction is equal to
(1) the...
(1) the...
Questions



Mathematics, 11.03.2020 11:04

Mathematics, 11.03.2020 11:04


Mathematics, 11.03.2020 11:05




Mathematics, 11.03.2020 11:09





English, 11.03.2020 11:12



Mathematics, 11.03.2020 11:16


Mathematics, 11.03.2020 11:22


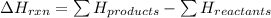
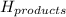 = Potential energy of the products
= Potential energy of the products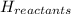 = Potential energy of the reactants
= Potential energy of the reactants