
Chemistry, 29.10.2019 00:31 ghostshadow1
For parts a and b, explain why the substances follow (or do not follow) the law of multiple proportions. take m(o) = 16 g/mol, m(mn) = 55 g/mol, and m(i) = 127 g/mol and derive the empirical formulae of all mentioned compounds. part a. sample a of manganese (mn) oxide contains 330 mg of mn and 144 mg of o, sample b — 330 mg of mn and 336 mg of o. part b. sample a of iodine (i) oxide contains 508 mg of i and 128 mg of o, sample b — 508 mg of i and 144 mg of o.
2. part a. calculate the mass of barium chloride dihydrate required to prepare a 1.500 l solution of 0.1500 mol/l ba2+. m(bacl2·2h2o) = 244.26 g/mol. part b. calculate the mass of copper(ii) sulfate pentahydrate required to prepare a 2.500 l solution of 0.2500 mol/l cu2+. m(cuso4·5h2o) = 249.69 g/mol.

Answers: 2
Another question on Chemistry

Chemistry, 23.06.2019 03:30
Ineed pls urgent 1-20 in order and fully detail step my step.
Answers: 1

Chemistry, 23.06.2019 08:30
Imagine you are a business executive who wants to pursue an environment policy for your company that limits pollution and uses fewer raw materials but would cost more what might be the discussion to your next broad meeting how would you make your case to your shareholders
Answers: 1

Chemistry, 23.06.2019 09:10
In a 28 g serving of cheese curls there are 247mg of sodium. how much sodium is in a 12.5 ounce bag
Answers: 1

Chemistry, 23.06.2019 10:10
Calculate the h3o+ concentration in a solution of acetic acid if the concentration of molecular acetic acid present at equilibrium is 9.97x10-3 m and k for the dissociation is 1.86x10-5. ch3cooh(aq)+h2o(l)+> h3o+(aq)+ch3coo-(aq) show me how to get the answer.
Answers: 3
You know the right answer?
For parts a and b, explain why the substances follow (or do not follow) the law of multiple proporti...
Questions

Mathematics, 09.12.2020 23:20

Mathematics, 09.12.2020 23:20

Mathematics, 09.12.2020 23:20

Spanish, 09.12.2020 23:20



English, 09.12.2020 23:20




English, 09.12.2020 23:20


Mathematics, 09.12.2020 23:20

Mathematics, 09.12.2020 23:20

Social Studies, 09.12.2020 23:20


Chemistry, 09.12.2020 23:20

Social Studies, 09.12.2020 23:30


Mathematics, 09.12.2020 23:30

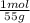 = 6x10⁻³ moles
= 6x10⁻³ moles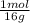 = 9x10⁻³ moles
= 9x10⁻³ moles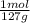 = 4x10⁻³ moles
= 4x10⁻³ moles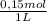 ×
×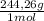 = 54,96g of BaCl₂.2H₂O
= 54,96g of BaCl₂.2H₂O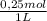 ×
×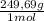 = 156,1g of CuSO₄.5H₂O
= 156,1g of CuSO₄.5H₂O

