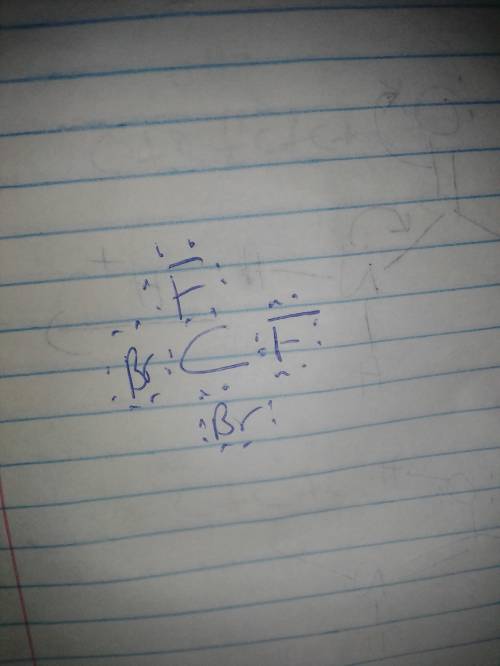Chemistry, 24.10.2019 21:43 ahmetagayew2004
Select all of the below statements that accurately describe the proper lewis structure for cbr2f2.- the formal charge on each bromine is -1.- there are 12 lone pairs of electrons on the molecule.- the formal charge on carbon is zero.- there are 4 covalent bonds within the molecule.- the formal charge on each fluorine is -1.

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 07:40
21. consider the following chemical reaction: n2+ o2 2 no if 10.0 g of n2 reacts with excess oxygen then how many grams of no can be formed? a) 10.7 g b) 21.4 g c) 32.9 g d) 42.8 g page 4 of 8
Answers: 2

Chemistry, 22.06.2019 08:30
In a chemical reaction at equilibrium, the rate of the forward reaction the rate of the reverse reaction. if the rate of the forward reaction more products are formed.
Answers: 1

Chemistry, 23.06.2019 01:30
Astudent states that 9.0 g of baking soda will form an unsaturated solution in 100 g of water. what do you need to know to decide whether this statement is correct? a. the temperature of the water and the molar mass of baking soda b. the percent by volume of the solution and the solubility of baking soda c. the temperature of the water and the solubility of baking soda at that temperature
Answers: 1
You know the right answer?
Select all of the below statements that accurately describe the proper lewis structure for cbr2f2.-...
Questions

Social Studies, 04.04.2021 18:00

Mathematics, 04.04.2021 18:00

Mathematics, 04.04.2021 18:00


Mathematics, 04.04.2021 18:00



English, 04.04.2021 18:00

History, 04.04.2021 18:00

English, 04.04.2021 18:00

Social Studies, 04.04.2021 18:00

Mathematics, 04.04.2021 18:00




Computers and Technology, 04.04.2021 18:10


Mathematics, 04.04.2021 18:10


Biology, 04.04.2021 18:10