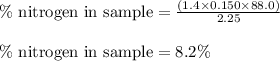The amount of nitrogen in an organic substance can be determined by an analytical method called the kjeldahl method, in which all the nitrogen in the organic substance is converted to ammonia. the ammonia, which is a weak base, can be neutralized with hydrochloric acid, as described by the equation nh3(aq)+hcl(aq)⟶nh4cl(aq) nh3(aq)+hcl(aq)⟶nh4cl(aq) if 88.0 ml88.0 ml of 0.150 m hcl(aq)0.150 m hcl(aq) is needed to neutralize all the nh3(g)nh3(g) from a 2.25 g sample of organic material, calculate the mass percentage of nitrogen in the sample.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 18:30
For each of the following mixtures decide if filtering would be suitable to separate the substances. explain your answers. oil in water sugar in water sand in water chalk in water tea leaves in a cup of tea
Answers: 2

Chemistry, 21.06.2019 21:50
H2so4(aq) + mg(s)—> mgso4(aq) +h2(g) which substance is the acid in the reaction?
Answers: 3


Chemistry, 22.06.2019 20:30
We are hoping to create 5.72 grams of glucose. the plant was given 4.75 liters of co2 and 2.81 g of h20. which reactant was the limiting reagent? how much excess mass did we have of the other reactant?
Answers: 2
You know the right answer?
The amount of nitrogen in an organic substance can be determined by an analytical method called the...
Questions

Computers and Technology, 25.05.2021 18:30

Arts, 25.05.2021 18:30



Mathematics, 25.05.2021 18:30

Physics, 25.05.2021 18:30



History, 25.05.2021 18:30

Mathematics, 25.05.2021 18:30

English, 25.05.2021 18:30

Mathematics, 25.05.2021 18:30


Geography, 25.05.2021 18:30


History, 25.05.2021 18:30


Chemistry, 25.05.2021 18:30


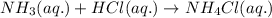
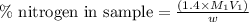
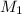 = molarity of acid used = 0.150 M
= molarity of acid used = 0.150 M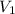 = Volume of acid used (in mL) = 88.0 mL
= Volume of acid used (in mL) = 88.0 mL