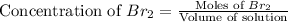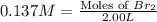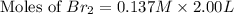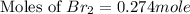Chemistry, 11.10.2019 03:00 simiyi1983
At elevated temperatures, molecular hydrogen and molecular bromine react to partially form hydrogen bromide: h2 (g) + br2 (g) ↔ 2hbr (g) a mixture of 0.682 mol of h2 and 0.440 mol of br2 is combined in a reaction vessel with a volume of 2.00 l. at equilibrium at 700 k, there are 0.516 mol of h2 present. at equilibrium, there are mol of br2 present in the reaction vessel.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 20:30
There is an area in idaho named craters of the moon where most of the ground is covered with basalt, adark gray, igneous rock with no visibl crystals. what can you infer about the geographical history of the area?
Answers: 1

Chemistry, 22.06.2019 06:30
Ineed someone to see if my answers are correct! if any are wrong let me know what the correct answers would be and how to get that answer! 1. how many moles of sodium chloride are in 28 grams od nacl? a. 265 mole naclb. 856 mole naclc. 479 mole of nacld. 1.2 mole nacl < my choice2. 734 grams of lithium sulfate (li2so4) are dissolved to make 2500 ml of solution what is rhe molaratiy? a. 2.67 mb. 4.56 mc. 3.89 m < my choiced. 1.78 m3. how many grams of cacl2 would be dissolved in 3.0 l of a 0.50 m solution of cacl2? a. 250 g cacl2 b. 166.5 g cacl2c. 113.65 g cacl2d. 98 g cacl2 < my choice4. suppose you had 58.44 g of nacl and you dissolved it in exactly 2.00 liters. the molarity if the solution would be 0.5 mtrue < my choicefalse 5. i would need 22g of naoh to make a 3.0 m solution using 250 ml of solvent.true < my choicefalse6. identify the solute: you have a .0195 m solution made from using 6.5 g of solute and 3 l of solvent. identify the solute by solving for molar weight.a. the solute is nacl because the molar weight is 58.43 g/mol < my choiceb. the solute is h2so4 because the molar weight is 98.06 g/molc. the solute is cacl2 because the molar weight is 111.11 g/mol
Answers: 1

Chemistry, 22.06.2019 08:30
Which of the following would have less momentum than a 52 kg cheetah running at 10 m/s?
Answers: 2

You know the right answer?
At elevated temperatures, molecular hydrogen and molecular bromine react to partially form hydrogen...
Questions




Mathematics, 11.03.2021 20:50

History, 11.03.2021 20:50

French, 11.03.2021 20:50

Mathematics, 11.03.2021 20:50

English, 11.03.2021 20:50


English, 11.03.2021 20:50



Mathematics, 11.03.2021 20:50


Physics, 11.03.2021 20:50


English, 11.03.2021 20:50

History, 11.03.2021 20:50

Social Studies, 11.03.2021 20:50

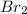 at equilibrium is 0.274 mole.
at equilibrium is 0.274 mole.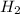 and
and 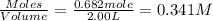
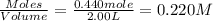
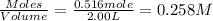
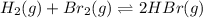
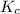 will be,
will be,![K_c=\frac{[HBr]^2}{[H_2][Br_2]}](/tpl/images/0308/9786/3b8cb.png)