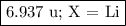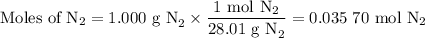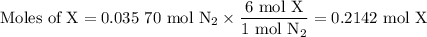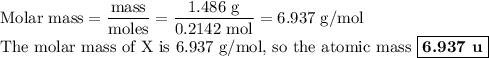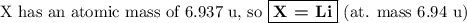Chemistry, 21.09.2019 07:30 MadiAbbott798
Nitrogen reacts with a metal to form a compound in which there are three atoms of the metal for each atom of nitrogen. if 1.486
g of the metal reacts with 1.000 g of nitrogen, what is the calculated atomic mass of the metal?
use your calculated atomic mass to identify the metal. (for your answer, input the proper chemical symbol for element x.)

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 06:40
Three alkali metals in group 1 are a. calcium, strontium, barium b. boron, aluminum, gallium c. sodium, potassium, rubidium d. fluorine, iodine, chlorine
Answers: 1

Chemistry, 22.06.2019 13:30
Table sugar completely dissolved in water is an example of a?
Answers: 1

Chemistry, 22.06.2019 15:30
Using the first volume and temperature reading on the table as v1 and t1, solve for the unknown values in the table below. remember to use the rules of significant figures when entering your numeric response.
Answers: 1

Chemistry, 22.06.2019 19:50
When the mercury level in a barometer decreases that atmospheric pressure has
Answers: 3
You know the right answer?
Nitrogen reacts with a metal to form a compound in which there are three atoms of the metal for each...
Questions

Mathematics, 30.08.2019 19:30



Geography, 30.08.2019 19:30



Mathematics, 30.08.2019 19:30

Social Studies, 30.08.2019 19:30

Mathematics, 30.08.2019 19:30

Health, 30.08.2019 19:30



Mathematics, 30.08.2019 19:30



English, 30.08.2019 19:30


Physics, 30.08.2019 19:30


English, 30.08.2019 19:30