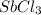Chemistry, 19.09.2019 20:30 milkshakegrande101
Abinary ionic compound is known to contain a cation with 51 protons and 48 electrons. the anion contains one-third the number of protons as the cation. the number of electrons in the anion is equal to the number of protons plus 1. what is the formula of this compound? what is the name of this compound?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 21:00
How many neutrons does an element have if its atomic number is 50 and its mass number is 166
Answers: 1

Chemistry, 22.06.2019 22:00
In order to complete this lab. you will need to be familiar with some common chemistry terms. complete the chemical change puzzle and list the relevant terms and their meaning below a.rectant b.product c.supernate
Answers: 3


Chemistry, 23.06.2019 06:30
How can the number of core electrons be determined from the periodic table
Answers: 1
You know the right answer?
Abinary ionic compound is known to contain a cation with 51 protons and 48 electrons. the anion cont...
Questions

English, 27.04.2021 22:20

Mathematics, 27.04.2021 22:20

Mathematics, 27.04.2021 22:20

Chemistry, 27.04.2021 22:20


Mathematics, 27.04.2021 22:20

History, 27.04.2021 22:20

Mathematics, 27.04.2021 22:20

History, 27.04.2021 22:20





Mathematics, 27.04.2021 22:20

Mathematics, 27.04.2021 22:20



Chemistry, 27.04.2021 22:20


English, 27.04.2021 22:20