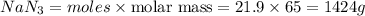Automotive air bags inflate when a sample of sodium azide nan3 is very rapidly decomposed.
2n...

Chemistry, 02.09.2019 22:30 danksans4912
Automotive air bags inflate when a sample of sodium azide nan3 is very rapidly decomposed.
2nan3 ? 2na +3n2
what mass of sodium azide is required to produce 2.6 feet3 (736 l) of nitrogen gas with the density of 1.25 g/l?

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 22:30
Will mark brainliest 26. which of these statements are true? (3 points) a. gases are compressible b. gases fill their containers completely c. the pressure of a gas is independent of the temperature d. gases have mass e. gases exert pressure f. the pressure of a gas is dependent on the volume g. gas pressure results from the collisions between gas particles h. gases have a definite volume and shape
Answers: 1

Chemistry, 22.06.2019 17:10
Increasing the substrate concentration in an enzymatic reaction could overcome which of the following? a) the need for a coenzymeb) allosteric inhibitionc) competitive inhibitiond) insufficient cofactors
Answers: 1

Chemistry, 22.06.2019 21:20
Phosgene (carbonyl chloride), cocl2, is an extremely toxic gas that is used in manufacturing certain dyes and plastics. phosgene can be produced by reacting carbon monoxide and chlorine gas at high temperatures: co(g) cl2(g)⇌cocl2(g) carbon monoxide and chlorine gas are allowed to react in a sealed vessel at 477 ∘c . at equilibrium, the concentrations were measured and the following results obtained: gas partial pressure (atm) co 0.830 cl2 1.30 cocl2 0.220 what is the equilibrium constant, kp, of this reaction
Answers: 2

Chemistry, 23.06.2019 03:30
Select the correct lewis structure for fluorine which is group 7a element?
Answers: 1
You know the right answer?
Questions








Computers and Technology, 30.11.2019 00:31


Computers and Technology, 30.11.2019 00:31





Computers and Technology, 30.11.2019 00:31


Mathematics, 30.11.2019 00:31

Mathematics, 30.11.2019 00:31


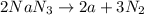

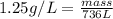

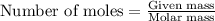
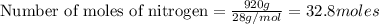
 are produced from= 2 mole of
are produced from= 2 mole of 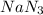
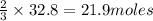 of
of