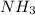Chemistry, 30.08.2019 01:30 natjojo0512
Consider the following balanced reaction between hydrogen and nitrogen to form ammonia: 3h2(g) + n2(g)→2nh3(g)how many moles of nh3 can be produced from 24.0 mol of h2 and excess n2? express the number of moles to three significant figures.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 02:20
Brainliesttt me asap! the below reaction can be categorized as more than one type of reaction. which reactions are these, and what are the types of reactions?
Answers: 1


Chemistry, 22.06.2019 21:00
Kp is the equilibrium constant for dissociation of the propionic acid dimer. what is the sign of the slope for a plot of the natural logarithm of kp vs. inverse temperature for this reaction?
Answers: 1

Chemistry, 22.06.2019 22:30
[ou.03jthe pictures below show the wavelengths and intensities of electromagnetic radiations emitted by three stars, star 1, star 2, and star 3. intensity intensity- intensity- 1000 3500 6000 8500 11000 wavelength (a) star 1 1000 3500 6000 8500 11000 1000 3500 6000 8500 11000 wavelength (a) wavelength (a) star 2 star 3 which of these statements is correct about the color of the three stars? star 2 is white in color o star 2 is yellow in color star 1 and star 3 are yellow in color star 1 and star 3 are white in color
Answers: 1
You know the right answer?
Consider the following balanced reaction between hydrogen and nitrogen to form ammonia: 3h2(g) + n2(...
Questions

Mathematics, 05.10.2021 16:50

Business, 05.10.2021 16:50

Physics, 05.10.2021 16:50


Computers and Technology, 05.10.2021 16:50

Mathematics, 05.10.2021 16:50


Biology, 05.10.2021 16:50

Social Studies, 05.10.2021 16:50

Biology, 05.10.2021 16:50

Social Studies, 05.10.2021 16:50

Computers and Technology, 05.10.2021 16:50





Health, 05.10.2021 16:50

Mathematics, 05.10.2021 16:50

English, 05.10.2021 16:50

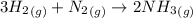
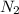 is present in large excess. It means that
is present in large excess. It means that 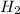 is the limiting reagent and the formation of the product is governed by
is the limiting reagent and the formation of the product is governed by