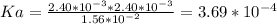Aspirin (acetylsalicylic acid, c9h8o4) is a weak monoprotic acid. to determine its acid-dissociation constant, a student dissolved 2.00 g of aspirin in 0.600 l of water and measured the ph. what was the ka value calculated by the student if the ph of the solution was 2.62?

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 08:00
An electron moved from shell n = 2 to shell n = 1. what most likely happened during the transition? a fraction of a photon was added. a photon of energy was absorbed. a fraction of a photon was removed. a photon of energy was released.
Answers: 1

Chemistry, 22.06.2019 22:30
Which one of the following bonds would you expect to be the most polar? a) b–h b) n–h c) p–h d) al–h e) c–h
Answers: 1

Chemistry, 23.06.2019 08:00
Amechanical wave that transports a lot of energy will have a
Answers: 2

Chemistry, 23.06.2019 10:40
Question 17 hydrogen is manufactured on an industrial scale by this sequence of reactions: +ch4gh2og ⇌ +cog3h2g k1 +cogh2og ⇌ +co2gh2g k2 the net reaction is: +ch4g2h2og ⇌ +co2g4h2g k write an equation that gives the overall equilibrium constant k in terms of the equilibrium constants k1 and k2. if you need to include any physical constants, be sure you use their standard symbols, which you'll find in the aleks calculator.
Answers: 2
You know the right answer?
Aspirin (acetylsalicylic acid, c9h8o4) is a weak monoprotic acid. to determine its acid-dissociation...
Questions

Physics, 13.07.2020 20:01


Mathematics, 13.07.2020 20:01

Mathematics, 13.07.2020 20:01

Mathematics, 13.07.2020 20:01

English, 13.07.2020 20:01

Mathematics, 13.07.2020 20:01

Mathematics, 13.07.2020 20:01


English, 13.07.2020 20:01

Chemistry, 13.07.2020 20:01

Mathematics, 13.07.2020 20:01

Mathematics, 13.07.2020 20:01

English, 13.07.2020 20:01

Mathematics, 13.07.2020 20:01



Mathematics, 13.07.2020 20:01


Social Studies, 13.07.2020 20:01

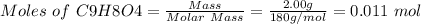
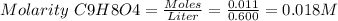
![Since\ pH = -log[H+]\\\\the \ [H+] = 10^{-pH } = 10^{-2.62} = 2.40*10^{-3} M](/tpl/images/0156/5580/28bcd.png)
![Ka = \frac{[H+][C9H7O4-]}{[C9H8O4]} = \frac{x^{2} }{0.018-x}](/tpl/images/0156/5580/1101d.png)