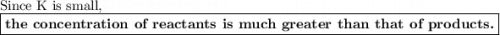Chemistry, 16.07.2019 03:10 19dansiste
The equilibrium-constant of the reaction no2(g)+no3(g)⇌n2o5(g) is k=2.1×10−20. what can be said about this reaction? a. at equilibrium the concentration of products and reactants is about the same. b. at equilibrium the concentration of products is much greater than the concentration of reactants. c. at equilibrium the concentration of reactants is much greater than that of products. d. there are no reactants left over once the reaction reaches equilibrium.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 03:00
Which step in naming unsaturated hydrocarbons is used for alkenes but not alkynes
Answers: 2



You know the right answer?
The equilibrium-constant of the reaction no2(g)+no3(g)⇌n2o5(g) is k=2.1×10−20. what can be said abou...
Questions













History, 22.07.2019 02:30

Social Studies, 22.07.2019 02:30


Chemistry, 22.07.2019 02:30

Computers and Technology, 22.07.2019 02:30



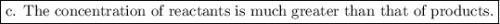
![K = \dfrac{[\text{Products}]}{[\text{Reactants}]}](/tpl/images/0094/8248/37418.png)