
Chemistry, 08.07.2019 23:40 sairaanwar67
For the reaction h2(g) + co2(g) ⇌ h2o(g) + co(g) at 700ºc, kc = 0.534. calculate the number of moles of h2 that are present at equilibrium if a mixture of 0.300 mole of co and 0.300 mole of h2o is heated to 700ºc in a 10.0-l container.

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 10:00
Miner's coal distributors does not mine coal itself, nor does it even store or handle the coal. instead, miner's solicits orders for low sulfur coal from other firms, then purchases the required amount from suppliers and directs them to ship the coal to its customers. what is miner's
Answers: 1


You know the right answer?
For the reaction h2(g) + co2(g) ⇌ h2o(g) + co(g) at 700ºc, kc = 0.534. calculate the number of moles...
Questions

Mathematics, 08.12.2020 01:00





Mathematics, 08.12.2020 01:00












Mathematics, 08.12.2020 01:00

Mathematics, 08.12.2020 01:00

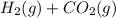 ⇄
⇄ 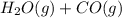 at 700ºC.
at 700ºC.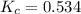
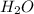 is heated to 700ºC.
is heated to 700ºC.![[H_2O]=\frac{300 mol}{10.0L}=30 M](/tpl/images/0067/3669/12641.png)
![H_2O=[H_2O]=\frac{300 mol}{10.0L}=30 M](/tpl/images/0067/3669/82ae5.png)
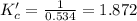
![K_c'=\frac{[H_2][CO_2]}{[CO][H_2O]}](/tpl/images/0067/3669/df550.png)
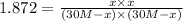
![[H_2]](/tpl/images/0067/3669/08a38.png)
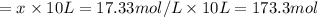
![[H_2]_{eq}=0.0173M](/tpl/images/0067/3669/87baf.png)
![Kc=\frac{[H_2O][CO]}{[H_2][CO_2]}](/tpl/images/0067/3669/4d854.png)
![[CO_2]_0=[H_2]_0=\frac{0.300mol}{10.0L} =0.030M](/tpl/images/0067/3669/89e63.png)
 due to the equilibrium, the law of mass action takes the following form:
due to the equilibrium, the law of mass action takes the following form: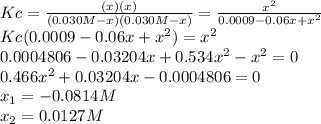
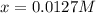
![[H_2]_{eq}=0.030M-0.0127M=0.0173M](/tpl/images/0067/3669/92f4c.png)



