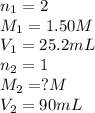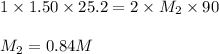Chemistry, 28.01.2020 11:31 covergurllaa
Avolume of 90.0 ml of aqueous potassium hydroxide (koh) was titrated against a standard solution of sulfuric acid (h2so4). what was the molarity of the koh solution if 25.2 ml of 1.50 m h2so4 was needed? the equation is 2koh(aq)+h2so4(aq)→k2so4(aq)+2h2o(l )

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 05:00
Cucl2 + 2nano3 cu(no3)2 + 2nacl what is the percent yield of nacl if 31.0 g of cucl2 reacts with excess nano3 to produce 21.2 g of nacl? 49.7% 58.4% 63.6% 78.7%
Answers: 1

Chemistry, 22.06.2019 21:30
In science class richard learns that a substance has a boiling point of 230 fahrenheit his teacher ask him to convert this temperature to degrees celsius what is the boiling point of his substance in degrees celsius
Answers: 3


Chemistry, 23.06.2019 06:30
An engineer decides to use a slightly weaker material rather than a stronger material, since she knows that the stronger material can break suddenly. this is an example of what? a choosing a material that will show warning before it fails b using composite materials that combine strength c using a material for multiple applications d using design techniques that increase efficiency and reduce cost
Answers: 3
You know the right answer?
Avolume of 90.0 ml of aqueous potassium hydroxide (koh) was titrated against a standard solution of...
Questions


Social Studies, 18.03.2021 02:10

Mathematics, 18.03.2021 02:10


Mathematics, 18.03.2021 02:10

Mathematics, 18.03.2021 02:10

Mathematics, 18.03.2021 02:10


Mathematics, 18.03.2021 02:10

Biology, 18.03.2021 02:10



Mathematics, 18.03.2021 02:10

Mathematics, 18.03.2021 02:10



Mathematics, 18.03.2021 02:10


Mathematics, 18.03.2021 02:10

Social Studies, 18.03.2021 02:10

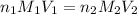
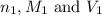 are the n-factor, molarity and volume of acid which is
are the n-factor, molarity and volume of acid which is 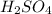
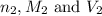 are the n-factor, molarity and volume of base which is KOH.
are the n-factor, molarity and volume of base which is KOH.