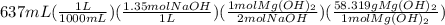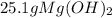Chemistry, 10.07.2019 07:30 andybiersack154
Sodium hydroxide and magnesium chloride react as shown by this equation: 2naoh + mgcl2 → mg(oh)2 + 2nacl. suppose the reaction begins with 637 milliliters of 1.35 m sodium hydroxide solution and excess magnesium hydroxide. what is the theoretical yield of magnesium hydroxide if the resulting solution has a volume of 2.82 liters? use the periodic table and the polyatomic ion resource. the mass of magnesium hydroxide formed is grams.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 07:40
22. a flask containing 450 ml of 0.50 m h2so4 was accidentally knocked to the floor. how many grams of nahco, do you need to put on the spill to neutralize the acid according to the following equation: h2so4(aq)+2 nahcos(aq) na,so(aq) +2 h20()+2 co2(g) d) 38 g a) 2.3 g b) 9.5 g c) 19 g
Answers: 1


Chemistry, 22.06.2019 13:00
6. using 3 – 4 sentences explain (in your own words) why water expands when it freezes? 7. using your knowledge of colligative properties explain whether sodium chloride or calcium chloride would be a more effective substance to melt the ice on a slick sidewalk. use 3 – 4 sentences in your explanation.
Answers: 1

Chemistry, 22.06.2019 22:30
What is a number added in front of a formula in order to balance the equation
Answers: 1
You know the right answer?
Sodium hydroxide and magnesium chloride react as shown by this equation: 2naoh + mgcl2 → mg(oh)2 +...
Questions

English, 06.11.2019 06:31





Biology, 06.11.2019 06:31

Health, 06.11.2019 06:31


History, 06.11.2019 06:31

Mathematics, 06.11.2019 06:31


Geography, 06.11.2019 06:31

English, 06.11.2019 06:31


Mathematics, 06.11.2019 06:31


Social Studies, 06.11.2019 06:31

Mathematics, 06.11.2019 06:31


Physics, 06.11.2019 06:31

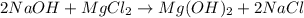
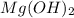 = 24.305 + 2( 15.999 + 1.008)
= 24.305 + 2( 15.999 + 1.008)