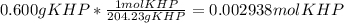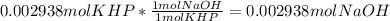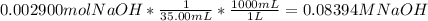Chemistry, 14.07.2019 12:00 potatogirl6811
Reagent grade potassium hydrogen phthalate (khp, mass 204.23g/mole) is a high molecular weight, stable, monoprotic solid acid. it is commonly used for standardizing sodium hydroxide solutions. what concentration of sodium hydroxide solution would be needed to titration 0.6000g of khp so that the volume of naoh needed is 35.00ml

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 00:20
What are the spectator ions in 2h+ + so42- + ca2+ + 2r → caso4 + 2h+ + 21?
Answers: 1

Chemistry, 22.06.2019 03:30
Adrop of acetone (nail polish remover) has a mass of 35 mg and a density of 0.788 g/cm3. what is its volume in cubic centimeters?
Answers: 3

Chemistry, 22.06.2019 14:30
100 grams of molten lead (600°c) is used to make musket balls. if the lead shot is allowed to cool to room temperature (21°c), what is the change in entropy (in j/k) of the lead? (for the specific heat of molten and solid lead use 1.29 j/g⋅°c; the latent heat of fusion and the melting point of lead are 2.45 × 104 j/kg and 327°c, respectively.)
Answers: 1

Chemistry, 22.06.2019 20:00
There are two steps in the usual industrial preparation of acrylic acid, the immediate precursor of several useful plastics. in the first step, calcium carbide and water react to form acetylene and calcium hydroxide: cac2 (s) + 2h2o (g) → c2h2 (g) + caoh2 (s) =δh−414.kj in the second step, acetylene, carbon dioxide and water react to form acrylic acid: 6c2h2 (g) + 3co2 (g) + 4h2o (g) → 5ch2chco2h (g) =δh132.kj calculate the net change in enthalpy for the formation of one mole of acrylic acid from calcium carbide, water and carbon dioxide from these reactions. round your answer to the nearest kj .
Answers: 3
You know the right answer?
Reagent grade potassium hydrogen phthalate (khp, mass 204.23g/mole) is a high molecular weight, stab...
Questions

History, 19.09.2019 16:10


Mathematics, 19.09.2019 16:10



Engineering, 19.09.2019 16:10


Health, 19.09.2019 16:10

Engineering, 19.09.2019 16:10


Computers and Technology, 19.09.2019 16:10






Biology, 19.09.2019 16:10



Computers and Technology, 19.09.2019 16:10