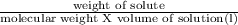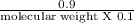Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 07:30
In a reaction (at equilibrium) that makes more moles of gas than it consumes, what is the effect of increasing the pressure?
Answers: 1

Chemistry, 22.06.2019 12:30
Sodium sulfate dissolves as follows: na2so4(s) → 2na+(aq) + so42- (aq). how many moles of na2so4 are required to make 1.0 l of solution in which the na concentration is 0.10 m?
Answers: 2

Chemistry, 22.06.2019 20:00
Nitrogen dioxide decomposes according to the reaction 2 no2(g) ⇌ 2 no(g) + o2(g) where kp = 4.48 × 10−13 at a certain temperature. if 0.70 atm of no2 is added to a container and allowed to come to equilibrium, what are the equilibrium partial pressures of no(g) and o2(g)
Answers: 2

Chemistry, 22.06.2019 23:30
Imagine a small synthetic vesicle made from pure phospholipids enclosing an interior lumen containing 1 mm glucose and 1 mm sodium chloride. if the vesicle is placed in pure water, which of the following happens faster? a. na+ diffuses out. b. cl– diffuses out. c. h2o diffuses in. d. glucose diffuses out. e. sodium chloride diffuses out.
Answers: 3
You know the right answer?
Asolution of 0.90 g of an unknown nonelectrolyte in 100 ml of water at 27◦c has an osmotic pressure...
Questions




Chemistry, 23.09.2020 14:01

Mathematics, 23.09.2020 14:01

Mathematics, 23.09.2020 14:01

History, 23.09.2020 14:01

Mathematics, 23.09.2020 14:01

Mathematics, 23.09.2020 14:01

Mathematics, 23.09.2020 14:01

Mathematics, 23.09.2020 14:01



Physics, 23.09.2020 14:01


Mathematics, 23.09.2020 14:01

History, 23.09.2020 14:01



History, 23.09.2020 14:01