
Biology, 18.12.2020 17:50 haimar8710
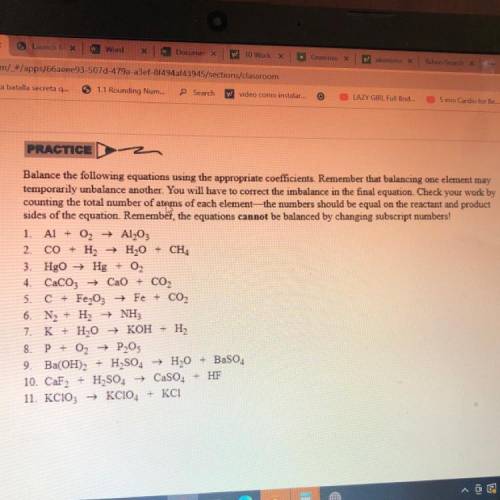
PRACTICE
Balance the following equations using the appropriate coefficients. Remember that balancing one element may
temporarily unbalance another. You will have to correct the imbalance in the final equation. Check your work by
counting the total number of atoms of each element—the numbers should be equal on the reactant and product
sides of the equation. Remember, the equations cannot be balanced by changing subscript numbers!
1. Al + O2 + Al2O3
2. CO + H2 + H2O + CH4
3. HgO + Hg + O2
4. CaCO3 + CaO + CO2
5. C + Fe2O3 → Fe + CO2
6. N2 + H2 + NH3
7. K + H2O → KOH + H2
8. P + O2 + P205
9. Ba(OH)2 + H2SO4 → H2O + BaSO4
10. CaF2 + H2SO4 → CaSO4 + HF
11. KCIO3 → KC1O4 + KCI

Answers: 1
Another question on Biology

Biology, 21.06.2019 20:00
Which of the following sex and generation combinations directly produces the pollen tube of angiosperms? a) male gametophyteb) female gametophytec) male sporophyted) female sporophyte
Answers: 1

Biology, 22.06.2019 03:50
The rapid decomposition of organic matter produces evidence which supports: the slow accumulation of coal deposits long ages of the earth rapid burial of vast amounts of vegetation biblical account of noah's flood
Answers: 2


Biology, 22.06.2019 09:20
Which statement explains how gravity and intertia work together
Answers: 1
You know the right answer?
PRACTICE
Balance the following equations using the appropriate coefficients. Remember that balancin...
Questions

Mathematics, 14.01.2021 23:10




Mathematics, 14.01.2021 23:10

Biology, 14.01.2021 23:10



Mathematics, 14.01.2021 23:10

History, 14.01.2021 23:10

Mathematics, 14.01.2021 23:10

Physics, 14.01.2021 23:10

Health, 14.01.2021 23:10





Geography, 14.01.2021 23:10

Mathematics, 14.01.2021 23:10



