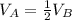Container A and container B hold samples of the same ideal gas. The volume and the pressure of container A is equal to the volume and pressure of container B, respectively. If Container A has half as many molecules of the ideal gas in it as Container B does, then which of the following mathematical statements is correct regarding the absolute temperatures TA and TB in Container A and Container B. respectively?
A. TA = TB/2.
B. TA = 4TB.
C. TA = TB/4.
D. TA = 2TB.
E. TA = TB

Answers: 1
Another question on Physics

Physics, 21.06.2019 17:20
Explain why the aperture of the spherical mirror should be small compared to the radius of curvature of the mirror.
Answers: 1


Physics, 22.06.2019 06:00
Imagine that someone pushes one marble toward a motionless marble. would there still be action-reaction forces involved in the collision? how might the marbles’ motions be changed? ?
Answers: 1

Physics, 22.06.2019 08:00
Who was the first scientist to measure speed as distance over time?
Answers: 1
You know the right answer?
Container A and container B hold samples of the same ideal gas. The volume and the pressure of conta...
Questions


Mathematics, 06.10.2020 15:01

History, 06.10.2020 15:01



Health, 06.10.2020 15:01



English, 06.10.2020 15:01



Mathematics, 06.10.2020 15:01





Biology, 06.10.2020 15:01



Mathematics, 06.10.2020 15:01