
Physics, 08.06.2021 01:00 gmoney1973
The liquid and gaseous state of hydrogen are in thermal equilibrium at 20.3 K. Even though it is on the point of condensation, model the gas as ideal and determine the most probable speed of the molecules (in m/s). What If? At what temperature (in K) would an atom of xenon in a canister of xenon gas have the same most probable speed as the hydrogen in thermal equilibrium at 20.3 K?

Answers: 1
Another question on Physics

Physics, 21.06.2019 21:20
Which of the following explains why it took so long for the public to accept the negative health effects of smoking? a)people were waiting for counter reasons, and those were presented.b)scientists had not completely finalized their results, which took 30 years.c)cigarettes were cheaply available and media channels were very inefficient.d)cigarette manufacturers were trying hard to bring an alternative into the market
Answers: 3

Physics, 22.06.2019 03:00
Ahot-air balloonist, rising vertically with a constant speed of 5.00 m/s releases a sandbag at the instant the balloon is 40.0 m above the ground. after it is released, the sandbag encounters no appreciable air drag. compute the velocity of the sandbag at 0.250 s after its release.
Answers: 2

Physics, 22.06.2019 04:50
The position of a crate sliding down a ramp is given by x=(0.05t^3) m, y=(1.7t^2) m, z=(6−0.85t^5/2) m, where t is in seconds. (a) determine the magnitude of the crate's velocity when t = 2 s. (b) determine the magnitude of the crate's acceleration when t = 2 s.
Answers: 2

Physics, 22.06.2019 09:10
Which lists the organs in the correct order as food passes from the mouth to anus?
Answers: 1
You know the right answer?
The liquid and gaseous state of hydrogen are in thermal equilibrium at 20.3 K. Even though it is on...
Questions

Mathematics, 14.07.2021 18:40


Biology, 14.07.2021 18:40




Chemistry, 14.07.2021 18:40

Mathematics, 14.07.2021 18:40

Mathematics, 14.07.2021 18:40

English, 14.07.2021 18:40

Biology, 14.07.2021 18:40


Mathematics, 14.07.2021 18:40



Physics, 14.07.2021 18:40


History, 14.07.2021 18:40


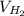 = √( 2RT /
= √( 2RT / 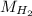 )
)

