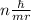Physics, 12.05.2021 01:00 payshencec21
The key insight that Bohr introduced to his model of the atom was that the angular momentum of the electron orbiting the nucleus was quantized. He introduced the postulate that the angular momentum could only come in quantities of nh/(2π), where h is Planck's constant and n is a nonnegative integer (0,1,2,3,…). Given this postulate, what are the allowable values for the velocity v of the electron in the Bohr atom? Recall that, in circular motion, angular momentum is given by the formula L= mvr.

Answers: 3
Another question on Physics

Physics, 21.06.2019 21:00
Apulley with a mechanical advantage of 5 will require you to pull times the amount of rope. a. 1/5 b. 5 c. 10 d. 15
Answers: 2

Physics, 21.06.2019 22:40
The desk has a weight of 75 lb and a center of gravity at g. determine the initial acceleration of a desk when the man applies enough force f to overcome the static friction at a and b. also, find the vertical reactions on each of the two legs at a and at b. the coefficients of static and kinetic friction at a and b are ms = 0.5 and mk = 0.2, respectively
Answers: 2

Physics, 22.06.2019 05:30
Agas expands from an initial volume of 0.040 m^3 and an initial pressure of 210 kpa to a final volume of 0.065 m^3 while its temperature is kept constant. how much work is done by the system?
Answers: 1

Physics, 22.06.2019 07:00
The cerebellum controls? thirst sensations sleep-wake cycles hand-eye coordination heart rate and pulse
Answers: 1
You know the right answer?
The key insight that Bohr introduced to his model of the atom was that the angular momentum of the e...
Questions

History, 18.03.2020 23:25




Mathematics, 18.03.2020 23:26

Mathematics, 18.03.2020 23:26

History, 18.03.2020 23:26


Mathematics, 18.03.2020 23:26

Biology, 18.03.2020 23:26

Social Studies, 18.03.2020 23:26

Mathematics, 18.03.2020 23:26



Mathematics, 18.03.2020 23:26


English, 18.03.2020 23:26