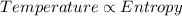Physics, 29.08.2019 00:30 kingkush85
Part a the third law of thermodynamics relates the entropy (randomness of matter to its absolute temperature. which of the following statements are in agreement with the third law of thermodynamics? check all that apply. check all that apply. at zero degrees kelvin, substances maximize entropy. at the melting point, the entropy of a system increases abruptly as the compound transforms into a liquid. as the temperature of a perfect crystal increases, the random vibrations of the molecules decrease. in a gaseous state, the entropy of the system is zero. in a crystalline state, there is a tendency to minimize entropy. at absolute zero, the interatomic distance within a crystal is minimized.

Answers: 2
Another question on Physics

Physics, 22.06.2019 02:30
Which of the following is not am example of a polymer? protein nylon kevlar concreta
Answers: 1

Physics, 22.06.2019 09:00
Atelevision set that has been running for a while heats up even the air around it. which two laws of thermodynamics could be used to analyze this scenario
Answers: 1


Physics, 22.06.2019 14:00
This is one type of line graph in which the independent variable is time plotted on the x axis and the dependent variable, also numerical, is plotted on the y axis.
Answers: 2
You know the right answer?
Part a the third law of thermodynamics relates the entropy (randomness of matter to its absolute tem...
Questions


Social Studies, 23.09.2019 15:30




Mathematics, 23.09.2019 15:30





Mathematics, 23.09.2019 15:30

Advanced Placement (AP), 23.09.2019 15:30


Mathematics, 23.09.2019 15:30

Chemistry, 23.09.2019 15:30

History, 23.09.2019 15:30

Mathematics, 23.09.2019 15:30

Mathematics, 23.09.2019 15:30


Chemistry, 23.09.2019 15:30