
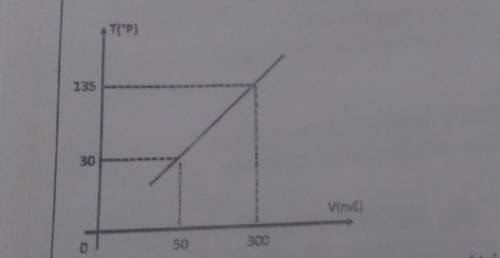
The diagram below shows the relationship between the volume v of a gas in a constant pressure gas thermometer and the corresponding temperature on an arbitrary T scale, calibrated in ° P
A) Determine the thermometric function for that device
B) Determine the temperature indicated by the device in ° P, when the gas volume is 1440ml
C) For a temperature of 1350 ° P, determine the new gas volume

Answers: 1
Another question on Physics

Physics, 22.06.2019 07:00
Within a pendulum, as potential energy decreases, energy increases. a. heat b. kinetic c. frictional d. gravitational
Answers: 1

Physics, 22.06.2019 10:50
Asheet of steel 1.5 mm thick has nitrogen atmospheres on both sides at 1200oc and is permitted to achieve a steady-state diffusion condition. the diffusion coefficient for nitrogen in steel at this temperature is 6 x 10- 11 m2 /s, and the diffusion flux is found to be 1.2 x 10-7 kg/m2 -s. also, it is known that the concentration of nitrogen in the steel at the high-pressure surface is 4 kg/m3 . how far into the sheet from this high-pressure side will the concentration be 2.0 kg/m3 ? assume a linear concentration profile.
Answers: 3

Physics, 22.06.2019 11:00
Consider a system to be two train cars traveling toward each other. what is the total momentum of the system before the train cars collide? kg • what must the total momentum of the system be after the train cars collide? kg •
Answers: 2

You know the right answer?
The diagram below shows the relationship between the volume v of a gas in a constant pressure gas th...
Questions


Advanced Placement (AP), 04.02.2021 07:10


Mathematics, 04.02.2021 07:10

Mathematics, 04.02.2021 07:10

Mathematics, 04.02.2021 07:10


Business, 04.02.2021 07:10


Mathematics, 04.02.2021 07:10






Biology, 04.02.2021 07:10

Mathematics, 04.02.2021 07:10



Mathematics, 04.02.2021 07:10



