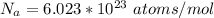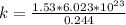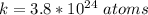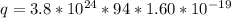Physics, 02.09.2020 23:01 Katmcfee7681
How many coulombs of positive charge (in units of 107 C) are there 1.53 kg of plutonium, given its atomic mass is 244 and that each plutonium atom has 94 protons? 1 atomic mass unit = 1.66*10-27 kg Enter a number with two digits behind the decimal point.

Answers: 1
Another question on Physics

Physics, 21.06.2019 20:30
F500 j of energy were added to 1 kg of each of these samples, which would experience the least temperature increase?
Answers: 2

Physics, 22.06.2019 05:50
Acylinder with a movable piston contains 11.7 moles of a monatomic ideal gas at a pressure of 1.32×10^5 pa. the gas is initially at a temperature of 300 k. an electric heater adds 43200 j of energy into the gas while the piston moves in such a way that the pressure remains constant. cp=20.79 j k^−1 mol^−1 for a monatomic ideal gas, and that the number of gas molecules is equal to avogadro's number (6.022×10^23) times the number of moles of the gas. (a) what is the temperature of the gas after the energy is added? (b) what is the change in volume of the gas? (c) how much work is done by the gas during this process?
Answers: 3

Physics, 22.06.2019 12:20
The diagram shows four locations in the electric field of a positive point charge m. at which location is the electric potential the greatest.
Answers: 2

Physics, 22.06.2019 16:00
Marvin records scientific data about lake superior. for which property has marvin forgotten to use an si unit of measurement? shore length high temperature low temperature annual precipitation
Answers: 3
You know the right answer?
How many coulombs of positive charge (in units of 107 C) are there 1.53 kg of plutonium, given its a...
Questions

Chemistry, 25.08.2019 03:30

Mathematics, 25.08.2019 03:30

Computers and Technology, 25.08.2019 03:30

Mathematics, 25.08.2019 03:30

English, 25.08.2019 03:30

Arts, 25.08.2019 03:30


Chemistry, 25.08.2019 03:30

Mathematics, 25.08.2019 03:30


English, 25.08.2019 03:30

Mathematics, 25.08.2019 03:30

Mathematics, 25.08.2019 03:30

History, 25.08.2019 03:30



English, 25.08.2019 03:30


History, 25.08.2019 03:30

Mathematics, 25.08.2019 03:30

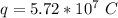
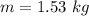
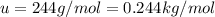
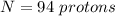
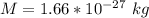
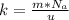
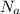 is the Avogadro constant with value
is the Avogadro constant with value