
The hydronium ion concentration in an aqueous solution is [H+] = 5.3 x10-4 M. What is the hydroxide ion concentration? Is this solution acidic or basic?
A.) [OH-] = 1.9 x10-11 M, acidic
B.) [OH-] = 4.7 x10-6 M, basic
C.) [OH-] = 1.1 x10-9 M, acidic
D.) [OH-] = 3.2 x10-8 M, acidic

Answers: 2
Another question on Physics

Physics, 22.06.2019 04:30
True or false when elements combine to form a mixture, the resulting chemical properties may be very different from those of the elements that make it.
Answers: 2

Physics, 22.06.2019 05:30
A2-kg bowling ball is 2.5 meters off the ground on a post when it falls. just before it reaches the ground, it is traveling 7 m/s. assuming that there is no air resistance, which statement is true?
Answers: 3


Physics, 22.06.2019 14:10
In one or two sentences, describe how you did in the balancing game. in a few more sentences, explain one strategy you learned for balancing more complex equations.
Answers: 2
You know the right answer?
The hydronium ion concentration in an aqueous solution is [H+] = 5.3 x10-4 M. What is the hydroxide...
Questions


Mathematics, 22.09.2020 14:01

Biology, 22.09.2020 14:01



Mathematics, 22.09.2020 14:01

Mathematics, 22.09.2020 14:01



Mathematics, 22.09.2020 14:01


Mathematics, 22.09.2020 14:01


Mathematics, 22.09.2020 14:01

Mathematics, 22.09.2020 14:01

Mathematics, 22.09.2020 14:01




Mathematics, 22.09.2020 14:01

![[H^+]=5.3\times 10^{-4}\ M](/tpl/images/0651/6541/426c9.png)
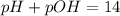
![pH=-\log [H^+]](/tpl/images/0651/6541/37e81.png)
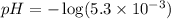
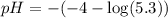
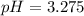
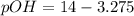
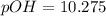
![pOH=-\log [OH^{-}]](/tpl/images/0651/6541/d7bcc.png)
![[OH]^{-1}=10^{-10.25}](/tpl/images/0651/6541/fc140.png)
![[OH]^{-1}=1.88\times 10^{-11}\ M](/tpl/images/0651/6541/93524.png)


