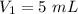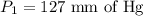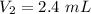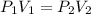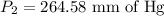A gas is placed in a 5.0-mL syringe. It exerts 127 mm Hg of pressure on the inside walls of the syringe. The syringe's plunger is pressed, reducing the volume of the syringe to 2.4 mL. The cap was not removed from the syringe, so none of the gas escapes. Assuming the temperature of the gas does not change, use Boyle's law to determine the pressure of the compressed gas.

Answers: 2
Another question on Physics

Physics, 21.06.2019 15:50
Aneutron and a proton combine to form a nucleus. why does the mass of the nucleus differ from the sum of the masses of the individual nucleons?
Answers: 1

Physics, 21.06.2019 17:20
Explain why the aperture of the spherical mirror should be small compared to the radius of curvature of the mirror.
Answers: 1

Physics, 21.06.2019 18:10
An insurance company wants to know if the average speed at which men drive cars is higher than that of women drivers. the company took a random sample of 27 cars driven by men on a highway and found the mean speed to be 72 miles per hour with a standard deviation of 2.2 miles per hour. another sample of 18 cars driven by women on the same highway gave a mean speed of 68 miles per hour with a standard deviation of 2.5 miles per hour. assume that the speeds at which all men and all women drive cars on this highway are both approximately normally distributed with unknown and unequal population standard deviations.
Answers: 2

Physics, 21.06.2019 21:30
Write the equation for momentum, first using symbols for the variables, then using words for the variables.
Answers: 2
You know the right answer?
A gas is placed in a 5.0-mL syringe. It exerts 127 mm Hg of pressure on the inside walls of the syri...
Questions

Mathematics, 01.09.2021 08:30

Mathematics, 01.09.2021 08:30

Computers and Technology, 01.09.2021 08:30





Mathematics, 01.09.2021 08:40

Physics, 01.09.2021 08:40


Social Studies, 01.09.2021 08:40


Mathematics, 01.09.2021 08:40

Mathematics, 01.09.2021 08:40

Mathematics, 01.09.2021 08:40

Social Studies, 01.09.2021 08:40

Social Studies, 01.09.2021 08:40

Mathematics, 01.09.2021 08:40

Chemistry, 01.09.2021 08:40

History, 01.09.2021 08:40