Atoms with a high electronegativity, like oxygen, have a strong attractive force
for electrons...

Physics, 24.04.2020 21:40 zairam2002
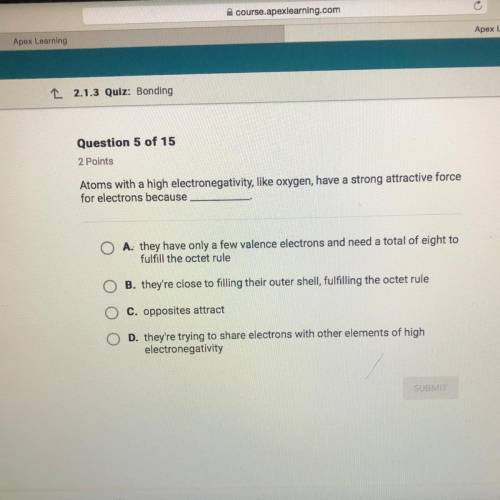
Atoms with a high electronegativity, like oxygen, have a strong attractive force
for electrons because
A. they have only a few valence electrons and need a total of eight to fulfill the octet rule
B. they're close to filling their outer shell, fulfilling the octet rule
C. opposites attract
D. they're trying to share electrons with other elements of high
electronegativity

Answers: 1
Another question on Physics

Physics, 22.06.2019 12:30
What is the power rating of the lightbulb if 3.0 a flow through it when connected to a 15 v battery
Answers: 1


Physics, 22.06.2019 17:30
Which of the choices are parts of an atom? select all that apply. a.) compound b.) electron c.) neutron d.) molecule e.) photon f.) proton g.) element
Answers: 1

Physics, 22.06.2019 23:50
What is part of a line has one endpoint and continues in one direction?
Answers: 1
You know the right answer?
Questions




Geography, 03.09.2020 01:01




Mathematics, 03.09.2020 01:01




Biology, 03.09.2020 01:01

Biology, 03.09.2020 01:01

Mathematics, 03.09.2020 01:01

History, 03.09.2020 01:01

Biology, 03.09.2020 01:01


Mathematics, 03.09.2020 01:01