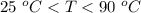Physics, 15.04.2020 17:46 abigail251
If you put a cup of hot chocolate at 90c on a table in a room kept at 25c you know it will cool down. How cool will the hot chocolate get?

Answers: 2
Another question on Physics

Physics, 22.06.2019 05:50
High schoolphysics 5+3 pts a neon light consists of a glass tube with metal wires at each end. when connected to a high-voltage source, the gas glows. if a fly lands on the glass tube, what will most likely happen? a) the fly will not feel a shock because the glass conducts any free electrons back into the gas. b) electrons will flow directly from the metal wires along the glass and shock the fly. c) the fly will not feel a shock because the glass insulates it from the electrons in the gas and the metal. d) electrons that are moving through the gas will be conducted through the glass and shock the fly.
Answers: 3

Physics, 22.06.2019 06:30
Which of these statements is true? a. quarks pair with leptons to form protons and neutrons. b. quarks pair with bosons to form photons. c. quarks pair with photons to form protons and neutrons. d. quarks pair with other quarks to form protons and neutrons. e. quarks pair with electrons to form protons.
Answers: 1

Physics, 22.06.2019 11:00
A2.00-m long piano wire with a mass per unit length of 12.0 g/m is under a tension of 8.00 kn. what is the frequency of the fundamental mode of vibration of this wire?
Answers: 3

Physics, 22.06.2019 13:10
The atoms in a nickel crystal vibrate as harmonic oscillators with an angular frequency of 2.3 × 1013 rad/s. the mass of a nickel atom is 9.75 × 10-26 kg. what is the difference in energy between adjacent vibrational energy levels of nickel? (h = 6.626 × 10-34 j • s, , 1 ev = 1.60 × 10-19 j)
Answers: 2
You know the right answer?
If you put a cup of hot chocolate at 90c on a table in a room kept at 25c you know it will cool down...
Questions


Mathematics, 06.11.2020 17:30

Arts, 06.11.2020 17:30


Geography, 06.11.2020 17:30




Mathematics, 06.11.2020 17:30

Mathematics, 06.11.2020 17:30



English, 06.11.2020 17:30

Mathematics, 06.11.2020 17:30

Mathematics, 06.11.2020 17:30

Biology, 06.11.2020 17:30

Mathematics, 06.11.2020 17:30

Social Studies, 06.11.2020 17:30


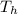 = 90 ⁰Croom temperature,
= 90 ⁰Croom temperature, 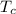 = 25 ⁰C.
= 25 ⁰C.