
In the classical view of the atom, Bohr pictured electrons orbiting the positively charged nucleus similar to how the planets orbit the Sun. While this picture was not entirely correct, it provides a good framework in which to make calculations about the energies of electrons. Different from the predictions of Newtonian mechanics, which allows any energy to be possible, Bohr described the electron orbits (now called orbitals) as having specific energies. Rank the following electron energy states according to their electron energies.

Answers: 3
Another question on Physics

Physics, 22.06.2019 05:10
A- a of 8.00 cm a of 34.0 nc. a , -, a of 25.0 nc is on of a of 15.0 cm .be toto aof ? a) b) c) j d) e)
Answers: 1

Physics, 22.06.2019 05:10
Total solidification times of three casting geometries are to be compared: a sphere, a cylinder, in which the length-to-diameter ration - 1.0, and a cube. volume = 1000 cm^3 for all three shhapes, and the same casting alloy is used. a) determine the solidification times of each geometry b) based on the results of part (a), which geometric element would make the best riser? c) if the mold constant = 3.5 min/cm^2 in chvorinov's rule, compute the total solidification time for each casting.
Answers: 3

Physics, 23.06.2019 04:00
Only 3% of the water on earth is usable by humans. even though that 3% is through the water cycle, we still need to be careful not to waste water.
Answers: 2

Physics, 23.06.2019 09:30
According to the second law of thermodynamics, what is true of entropy in all natural systems? a.it will always decrease. b.it will always increase. c.it will always remain the same. d.it is impossible to measure. e.it will always equal zero.
Answers: 1
You know the right answer?
In the classical view of the atom, Bohr pictured electrons orbiting the positively charged nucleus s...
Questions


Mathematics, 27.07.2020 02:01

SAT, 27.07.2020 02:01


Mathematics, 27.07.2020 02:01



History, 27.07.2020 02:01





Mathematics, 27.07.2020 02:01

Mathematics, 27.07.2020 02:01

Advanced Placement (AP), 27.07.2020 02:01

Mathematics, 27.07.2020 02:01

Mathematics, 27.07.2020 02:01



 Second excited state (n=3)
Second excited state (n=3) 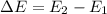 = hv, where h = planck's constant.
= hv, where h = planck's constant.

