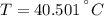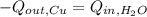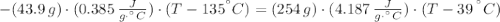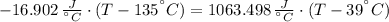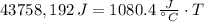Physics, 13.03.2020 04:59 silvernekoo
A 43.9-g piece of copper (CCu= 0.385 J/g°C) at 135.0°C is plunged into 254 g of water at 39.0°C. Assuming that no heat is lost to the surroundings, what will the final temperature of the system be?

Answers: 1
Another question on Physics


Physics, 22.06.2019 11:20
The ultracentrifuge is an important tool for separating and analyzing proteins. because of the enormous centripetal accelerations, the centrifuge must be carefully balanced, with each sample matched by a sample of identical mass on the opposite side. any difference in the masses of opposing samples creates a net force on the shaft of the rotor, potentially leading to a catastrophic failure of the apparatus. suppose a scientist makes a slight error in sample preparation and one sample has a mass 10 mg larger than the opposing sample. if the samples are 12 cm from the axis of the rotor and the ultracentrifuge spins at 70,000 rpm, what is the magnitude of the net force on the rotor due to the unbalanced samples? ( be thorough on your answer)
Answers: 3

Physics, 22.06.2019 22:40
Calculate the volume of this regular solid. what is the volume of the cone? round your answer to the nearest hundredth
Answers: 1

You know the right answer?
A 43.9-g piece of copper (CCu= 0.385 J/g°C) at 135.0°C is plunged into 254 g of water at 39.0°C. Ass...
Questions




Mathematics, 16.10.2020 16:01



Computers and Technology, 16.10.2020 16:01

Chemistry, 16.10.2020 16:01




Spanish, 16.10.2020 16:01

English, 16.10.2020 16:01

Physics, 16.10.2020 16:01

Chemistry, 16.10.2020 16:01


Mathematics, 16.10.2020 16:01


Mathematics, 16.10.2020 16:01