
Temperature plays in important part in most chemical reactions. Generally speaking, the higher the temperature, the higher the rate of reaction due to the increased amount of energy available as well as an increase in particle contact.
Suppose a chemical reaction was discovered in which temperature was not relevant at all. An experiment was devised where all other factors were allowed to vary - pressure, number of particles, etc. - and the temperature was increased from 10°C to 50°C while the rate of the reaction was measured.
How would the graph seen here change with respect to this new reaction?
A) The graph would show a random scatter of data points.
B) The graph would be a single vertical line at some temperature.
C) The graph would be a single horizontal line at some reaction rate
D) The graph would not be significantly different from the one shown.
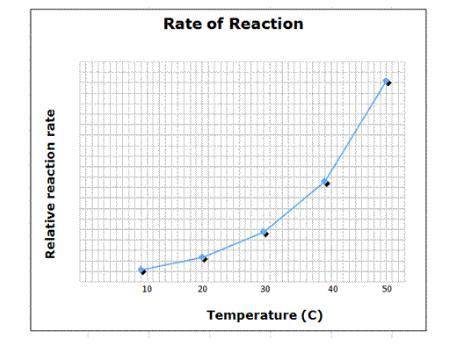

Answers: 3
Another question on Physics

Physics, 22.06.2019 07:30
Examine the nuclear reacti why is this classified as a nuclear reaction rather than a chemical reaction? it is not balanced. a new compound is formed. a change has occurred in a nucleus. a new element has been formed.
Answers: 2

Physics, 22.06.2019 09:00
What is a possible result of higher air temperature caused by global warming
Answers: 1

Physics, 22.06.2019 17:30
Atruck driver is attempting to deliver some furniture. first, he travels 8 km east, and then he turns around and travels 3 km west. finally, he turns again and travels 13 km to his destination. what is the drivers distance
Answers: 1

You know the right answer?
Temperature plays in important part in most chemical reactions. Generally speaking, the higher the t...
Questions


English, 10.11.2021 22:30







Mathematics, 10.11.2021 22:30


Spanish, 10.11.2021 22:30

Chemistry, 10.11.2021 22:30






Mathematics, 10.11.2021 22:30


Mathematics, 10.11.2021 22:30




