
How much heat energy is required to convert 93.4 g of solid ethanol at − 114.5 ° c to gasesous ethanol at 149.8 ° c ? the molar heat of fusion of ethanol is 4.60 kj/mol , and its molar heat of vaporization is 38.56 kj/mol . ethanol has a normal melting point of − 114.5 ° c and a normal boiling point of 78.4 ° c . the specific heat capacity of liquid ethanol is 2.45 j / g ⋅ ° c , and that of gaseous ethanol is 1.43 j / g ⋅ ° c .

Answers: 3
Another question on Physics

Physics, 22.06.2019 15:50
An object with initial temperature 130 ∘ f is submerged in large tank of water whose temperature is 50 ∘ f . find a formula for f ( t ) , the temperature of the object after t minutes, if the cooling constant is k = − 0.2 . remember newton's law of cooling (the rate of change of temperature with respect to time is equal to k times the difference between the temperature of the object and the surrounding temperature) ! : )
Answers: 1

Physics, 23.06.2019 00:30
Why does the equilibrium position of the spring change when a mass is added to the spring? will the mass oscillate around the new equilibrium position of the spring or the previous position without a mass attached to the spring? if the equilibrium position of the spring changes by 20 cm (assuming no initial mass) when a mass is added to the spring with constant 4.9 kg/s^2, what is the mass of the object attached to the spring?
Answers: 2

Physics, 23.06.2019 08:10
Ais a multilevel way of grouping items based on common properties among them. a. memory b. conceptual hierarchy c. schema d. semantic network
Answers: 1

Physics, 23.06.2019 13:30
What feature will form as a result of the movement displayed in this illustration? a.convergent boundaryb.mid-ocean ridgec.deep-sea trenchd.transform boundary
Answers: 1
You know the right answer?
How much heat energy is required to convert 93.4 g of solid ethanol at − 114.5 ° c to gasesous ethan...
Questions


Chemistry, 19.01.2021 07:00


Biology, 19.01.2021 07:00

Mathematics, 19.01.2021 07:00

Chemistry, 19.01.2021 07:00

Chemistry, 19.01.2021 07:00



Chemistry, 19.01.2021 07:00

Chemistry, 19.01.2021 07:00




Mathematics, 19.01.2021 07:00

Computers and Technology, 19.01.2021 07:00

Social Studies, 19.01.2021 07:00

Mathematics, 19.01.2021 07:00

English, 19.01.2021 07:00

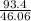 = 2.02 moles
= 2.02 moles

