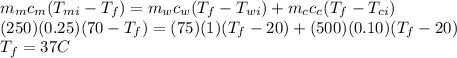Physics, 20.12.2019 18:31 naeaamm6981
Ahot (70°c) lump of metal has a mass of 250 g and a specific heat of 0.25 cal/g⋅°c. john drops the metal into a 500-g calorimeter containing 75 g of water at 20°c.
the calorimeter is constructed of a material that has a specific heat of 0.10 cal/ g⋅°c.
when equilibrium is reached, what will be the final temperature? cwater = 1.00 cal/g⋅°c.
a. 114°cb. 72°cc. 64°cd. 37°c

Answers: 3
Another question on Physics



Physics, 22.06.2019 22:30
Find v1, the speed of the fluid in the left end of the main pipe. express your answer in terms of h1, h2, g, and either a1 and a2 or γ, which is equal to a1a2.
Answers: 2

Physics, 22.06.2019 23:20
Consider a production line with five stations. station 1 can produce a unit in 9 minutes. station 2 can produce a unit in 10 minutes. station 3 has two identical machines, each of which can process a unit in 12 minutes (each unit only needs to be processed on one of the two machines. station 4 can produce a unit in 5 minutes. station 5 can produce a unit in 8 minutes. which station is the bottleneck station?
Answers: 2
You know the right answer?
Ahot (70°c) lump of metal has a mass of 250 g and a specific heat of 0.25 cal/g⋅°c. john drops the m...
Questions

Mathematics, 02.10.2019 23:00



Health, 02.10.2019 23:00


Geography, 02.10.2019 23:00

Social Studies, 02.10.2019 23:00

History, 02.10.2019 23:00


History, 02.10.2019 23:00


Mathematics, 02.10.2019 23:00


English, 02.10.2019 23:00




Mathematics, 02.10.2019 23:00


Biology, 02.10.2019 23:00

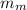 = mass of lump of metal = 250 g
= mass of lump of metal = 250 g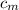 = specific heat of lump of metal = 0.25 cal/g°C
= specific heat of lump of metal = 0.25 cal/g°C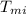 = Initial temperature of lump of metal = 70 °C
= Initial temperature of lump of metal = 70 °C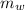 = mass of water = 75 g
= mass of water = 75 g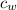 = specific heat of water = 1 cal/g°C
= specific heat of water = 1 cal/g°C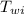 = Initial temperature of water = 20 °C
= Initial temperature of water = 20 °C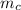 = mass of calorimeter = 500 g
= mass of calorimeter = 500 g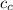 = specific heat of calorimeter = 0.10 cal/g°C
= specific heat of calorimeter = 0.10 cal/g°C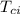 = Initial temperature of calorimeter = 20 °C
= Initial temperature of calorimeter = 20 °C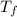 = Final equilibrium temperature
= Final equilibrium temperature